Do clinical trials for cancer exclude some patients?
Clinical trials for cancer are crucial for developing effective treatments to help cancer patients. Many people volunteer to take part in the trials, but not everyone is accepted. Read on to find out why some people are excluded from the trials and what happens next.
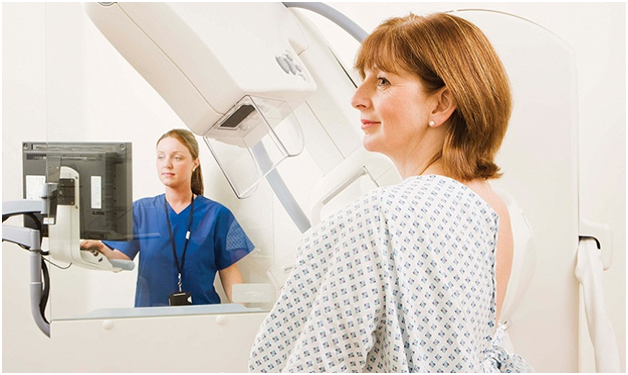
Sometimes, people with cancer try to take part in a clinical trial for a new treatment, but the clinic won’t accept them onto the trial. There are many reasons why not. Trials have strict eligibility requirements to ensure the correct patients are chosen for the study.
Eligibility requirements
Some requirements include age and the stage of the patient’s cancer at the time of the trial. People have tests to determine if they fit the strict criteria for the trial. For example, if blood tests show raised or lowered levels of certain chemicals or indicators of organ function that are outside the required range, the patient will be excluded from the trial.
Patients sometimes have to keep looking for trials that suit their own stage of the disease and physical condition. Sometimes, trials are very strict, but others are more lenient. The strict requirements can be a huge barrier for cancer patients, as for many, this is their only hope of finding an effective treatment, if they have exhausted other options.
You can find out how to take part in clinical trials by asking your doctor’s advice on participation. There are also patient organisations that can advise you. Also, you can get advice online from expert services such as the NHS.
Other options for cancer patients
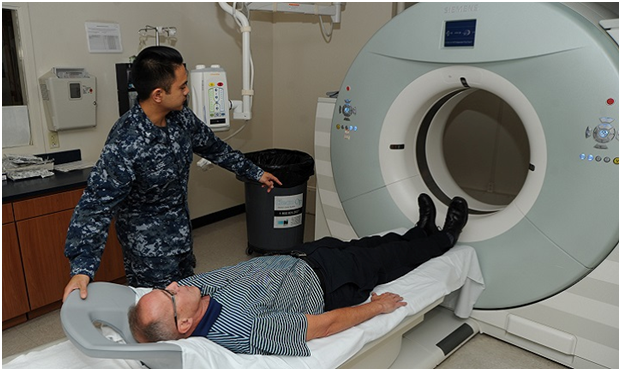
When clinics run Adaptive Phase 1 Studies, they have to ensure the right type of people is taking part. Pharmacologists from top clinics, such as Richmond Pharmacology have to check all the criteria of each patient to ensure they are at the right stage of the disease and meet the requirements regarding health and age. If the wrong people are accepted, this could hinder the trial or make the results misleading.
Many health experts believe the trials are often too selective. The participation rate in cancer trials is 3 percent. However, they need to be selective in some instances to ensure the reliability of the trial data. For example, HIV patients who also have cancer are always ruled out of trials, as are patients who are under 18.